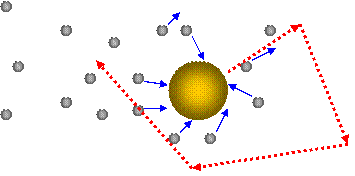
 He was examining plant seeds and was puzzled by their movement even when they were dead. The Scottish botanist Robert Brown was the first one known to investigate Brownian motion in 1827. The beads each have a random trajectory and do not all move in the same direction. The short movie above shows fluorescent beads moving through a solution. Fluorescent beads can be seen moving in solution. The individual particles are tracked by detecting the photons or light released by single molecules as they move through the solution. Particles move away from their original position and randomly distribute.Ī common experiment where Brownian movement can easily be tracked is watching fluorescent dyes in a solution. All particles in a liquid or gas are moving due to Brownian motion.ĭiffusion happens in part due to Brownian motion. Examples of Brownian Motionīrownian motion can be hard to observe. The effect of all the smaller particles hitting the larger particles is enough to counteract gravity and cause the large particles to stay in solution. Instead, Brownian movement is what keeps the particles in solution. The larger particles stay suspended in solution and do not settle out to the bottom as predicted due to gravity. Milk and fog are common examples of colloids. Therefore, smaller particles will move faster and travel further than larger particles.Ī colloid is a homogeneous mixture with large particles suspended in a solution of another substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
